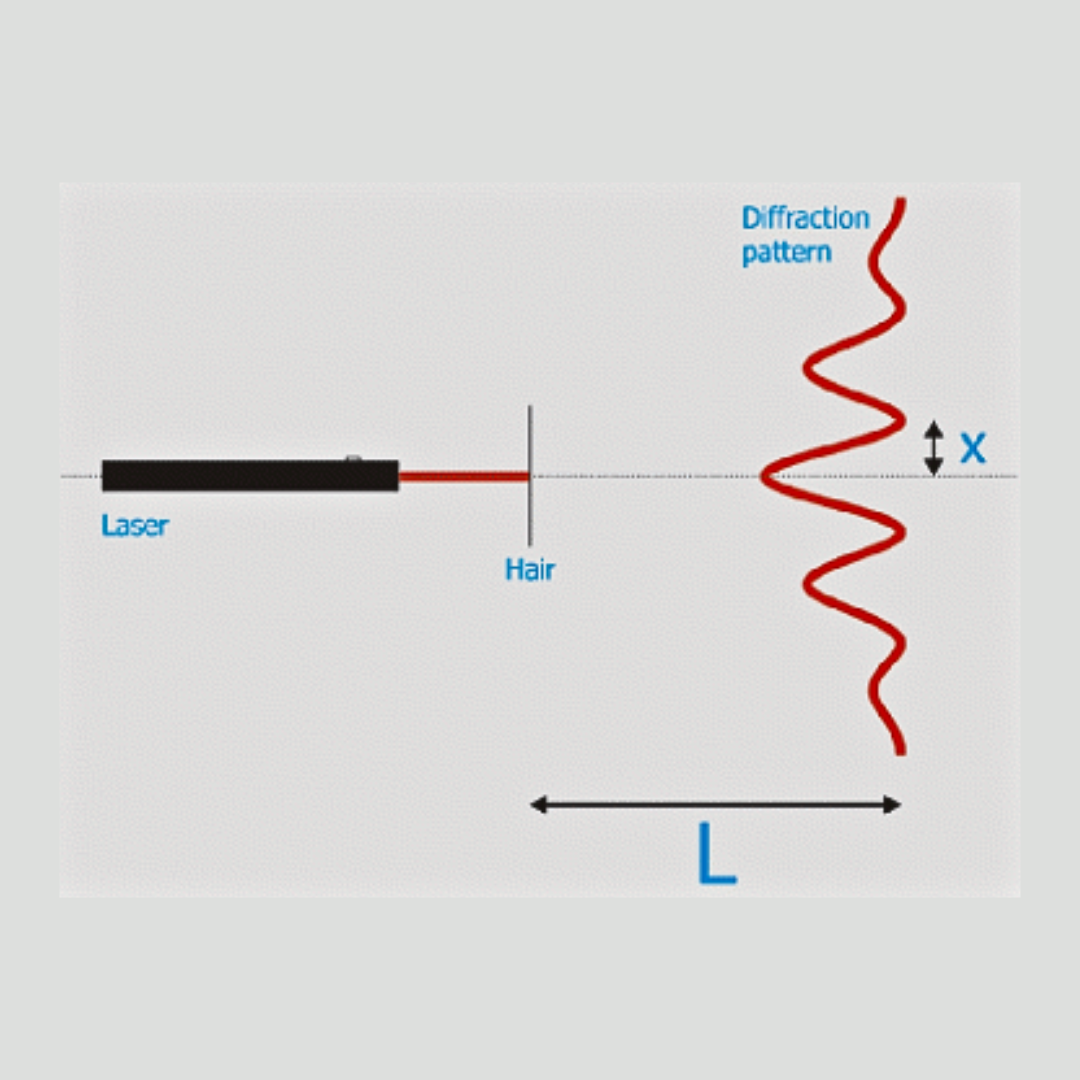
Lasers, Light and Optics - Free Lesson Downloads
Welcome to Laser Classroom’s Free Light, Laser and Optics Lessons and Experiments!! Everything here illuminates the naturally STEM integrated topics of light, lasers and optics with step by step instructions for engaging your students.
The free downloadable optics lessons on this page bring abstract concepts into focus and introduce the amazing science, technology, engineering and math of the basics like reflection, refraction and diffraction as well as applications like spectroscopy, invisibility and energy. Most even have printable activity worksheets for the students!
Each page below gives a preview of the lesson. To download the full lessons enter your email here and we'll send you a .ZIP file with everything you need!
Download free lessons
We'll email you a link to download the .ZIP file with over two dozen PDF lessons!
Free LightBlox lessons

Light Blox - LED color mixing educational kit
To make the experiments above fun and easy, use our blue, green, and red LEDs for DIY color mixing, reflection and refraction experiments!







Free LaserBlox and Laser Pointer Lessons







LaserBlox Educational Laser Pointers
To safely do the above lessons, consider LaserBlox! Designed for safety and education, our violet, green, and red laser pointers take optics learning to the next level.